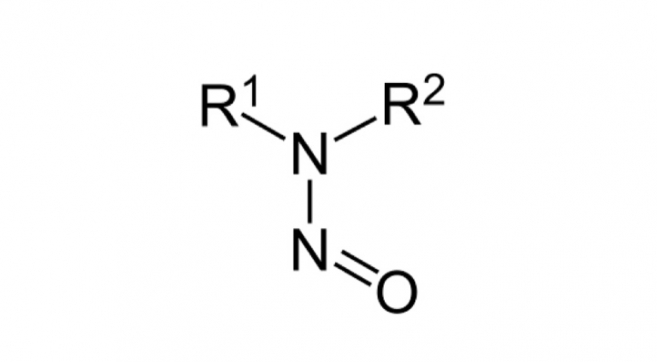
Questionnaire for Excipient Nitrosamines Risk Evaluation
Category: IPEC EuropeIPEC Europe developed a questionnaire in consultation with other pharmaceutical-related associations (including EFPIA) to facilitate gathering information on pharmaceutical excipients for drug product manufacturers to perform nitrosamine risk evaluations, required by several regulatory authorities globally.
IPEC Europe strongly encourages both excipient manufacturers and their drug product manufacturer customers to use this standard format. This will greatly facilitate timely data collection from excipient manufacturers enabling a more efficient process of conducting the said risk assessments.
IPEC Europe welcomes any feedback on the form (info@ipec-europe.org).
---
A new version of the questionnaire is now available : click here